Louis Garguilo
ARTICLES BY LOUIS
-
9/23/2021
You are an emerging biopharma outsourcing to a trusted CDMO that has selected its location wisely – an important factor in your selecting it. Now you’ve determined to establish some of your own development or manufacturing facilities. Would it be beneficial to build close to your CDMO?
-
9/16/2021
With a more flexible and relational approach, CDMOs and emerging drug sponsors can end up putting together a different contract altogether – one driving a new pricing and calculation that works for both sides. Here's details to one example.
-
9/13/2021
Somebody has to set the strategic integration of in-house development and manufacturing with external activities; identify for investors why that is the wisest strategy; explain why the CDMO selected is the best fit. Those somebodies are CEOs. Here’s more from ISR Reports.
-
9/8/2021
What's really in it for your independent advisors and board members? We've some firsthand answers that will catalyze some thoughts on how you receive potentially invaluable advice from a trusted executive-level mentor, advisor, and additive governing voice – and not only regarding outsourcing.
-
9/7/2021
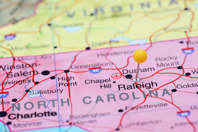
In the beginning, there were Big Pharma concentrations. Decades later, biotech centers emerged. Now we have our first CDMO cluster. It’s in North Carolina. And it’s worth giving this some thought if you are outsourcing drug development and manufacturing.
-
9/1/2021
When outsourcing drug development and manufacturing, how involved should the CEOs of emerging biopharma be in the requisite day-to-day activities and processes? James Mackay, CEO of Aristea Therapeutics, and independent board member to emerging biopharma, says: “The CEO is likely to be the ultimate decision-maker on more activities related to outsourcing than they might expect.” Here are some best practices.
-
8/20/2021
Today drug development and manufacturing outsourcing is a ubiquitous business-enabling activity to gain entrée to advanced technologies and capabilities. Those are the capabilities upon which your programs and overall organization are centered. Are you, then, outsourcing your future?
-
8/18/2021
A discussion on lessons learned from the pandemic featured Remo Colarusso, Vice President, Janssen Supply Chain, Johnson & Johnson, and Ken Kent, Senior Vice President, Chemical Development and Manufacturing Operations, Gilead Sciences, at a recent DCAT Virtual Week event.
-
8/16/2021
The fundamental challenge in today’s drug development and manufacturing outsourcing space is a misaligned market. CDMOs are getting bigger; emerging drug developers are getting smaller. Something has to give. Actually, that give has begun.
-
8/11/2021
James Mackay, President & CEO, Aristea Therapeutics, serves as an independent board member to two other San Diego-based biopharma companies, and one in Massachusetts. What exactly is his role? What advice does he give on outsourcing? And should you have an independent board member as well?