Hospitals To Serve Up IVs, And Now APIs?

By Louis Garguilo, , Chief Editor, Outsourced Pharma Follow Me On Twitter @Louis_Garguilo

Outsourced Pharma readers must be scratching your heads over the mass-media reported news splash that Intermountain Healthcare is leading four U.S.-based “health care systems” in a consortium to form a generic drug company.
In other words, hospitals are getting into APIs. And this, we are told, will save the world from shortages, while reducing pricing for generic drugs.
Oh, and by the way, the organization established to facilitate these feats will be “non-profit.”
Of course the mass media dove as deeply into this as into a three-inch stream. Let’s just say some questions remain, such as: “How’s that all going to work?”
Gil Roth, Founder and President, Pharma and Biopharma Outsourcing Association (PBOA), also appears intrigued by this lack of investigatory fortitude. He tweeted:
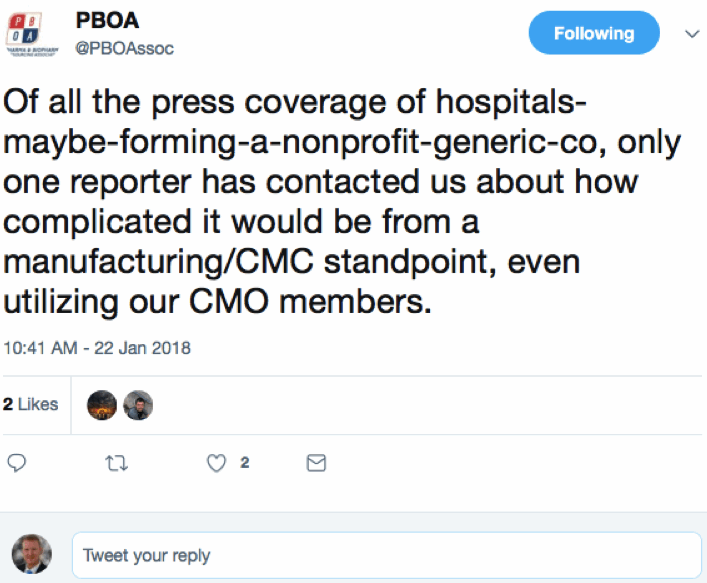
Actually, one of a mix of reactions when I first encountered this news was that there could be an opportunity for CDMOs somewhere in this. But just as those thoughts were germinating, GlobalData placed a press release in my inbox titled, “Hospital-owned generics initiative unlikely to benefit the CMO industry,” and quoting a person we are very familiar with in these pages, Jim Miller:
“A recently-announced plan to establish a not-for-profit generic pharmaceutical company is unlikely to be a boon to the contract manufacturing organization (CMO) industry ... A consortium of five hospital organizations representing 450 hospitals aims to address the problem of needed medications that are in short supply or have undergone extraordinary price increases. The consortium is led by Intermountain Healthcare and includes the US Department of Veterans Affairs’ Veterans Health Administration, SSM Health, Trinity Health, and Ascension … Jim Miller, President and Founder of PharmSource, a GlobalData company, comments: ‘Our initial reaction is that the group has some significant hurdles to overcome, the most serious of which is the manufacturing capacity to produce the drugs. Constraints on good manufacturing practices (GMP)-compliant capacity have been a significant contributor to the product shortages the new initiative seeks to address.’”
“Furthermore,” the release continues, “the hospital-owned generics company will face a difficult decision: it can either build sophisticated procurement, quality assurance, and distribution operations capable of creating and managing a global network of active pharmaceutical ingredient (API) and drug product suppliers, or it can build a manufacturing facility that will require a sizeable investment (easily $50 million at the scale they are addressing) and 3–5 years to complete.
“It’s also worth noting that none of the advisory committee members named in the hospital group’s press release has any significant generic industry experience … One is led to question whether the group really understands what it is getting into … a more likely scenario, at least in the near term, is that the hospital group evolves into a purchasing consortium that negotiates contracts with existing generics companies guaranteeing pricing and volumes sufficient to maintain a stable supply of the products currently in short supply.”
For the record, I’d only slightly disagree with the above analysis regarding the advisory group. Two former Amgen officials on the board could prove significant, signal more direct pharma involvement, and lends some immediate credibility to the staging of this generics “start-up.”
An Avalanche Of Adjectives
Let’s look at the specific mission statement and industry status in the press release from Intermountain Healthcare:
“The new company intends to be an FDA approved manufacturer and will either directly manufacture generic drugs or sub-contract manufacturing to reputable contract manufacturing organizations, providing patients an affordable alternative to products from generic drug companies whose capricious and unfair pricing practices are damaging the generic drug market and hurting consumers. The company will also seek to stabilize the supply of essential generic medications administered in hospitals, many of which have fallen into chronic shortage.”
Reputable. Affordable. Capricious. Unfair. Damaging. Hurting. Chronic.
Have we got all that?
And in case anyone was wondering, as reported in a Wall Street Journal article (Hospitals Plan to Produce Generic Drugs in Bid to Fix Troubled Market), Intermountain President and Chief Executive Marc Harrison is quoted as saying: “We have no designs on being a major pharmaceutical power.”
Well, glad that’s cleared up.
Along with this last rhetorical flourish of anti-hyperbolic hyperbole, Dr. Harrison does get down to basic tenets in that WSJ article. “This is a very specific problem we’re seeking to solve with a very specific solution,” he says, and the new company will “provide an alternative to sole-sourced, overpriced, in-short-supply medications.”
Cutting through the adjectives for a moment, what we have here is an attempt at innovation targeted at the drug supply chain. With the hundreds of articles I’ve included in these pages extolling the search for innovation in drug development and manufacturing outsourcing, I have to question my own questioning of this initiative.
Perhaps holding me back is that final little detail, which appears largely unstudied so far: the “non-profit” designation of the new organization. A “non-profit” pharma organization (whatever that may mean) established by and directly feeding gigantic and ever-growing “health care systems” doesn’t ring a clear bell here. In fact, it sounds an alarm.
