Characterizing Novel Microparticle-Encapsulated Drug Formulations Part 2
By Trae Carroll, Associate Pharmaceutical Scientist

Part 2 of a 2 part series
Microparticle encapsulated drugs are increasingly desirable dosage forms as clinicians seek to target specific sites in the body for controlled release therapy. In the previous blog post, we addressed the challenge of successfully formulating such compounds for oral administration. However, once you have successfully encapsulated an active pharmaceutical ingredient (API), the next task is to characterize that product and understand its release kinetics, such that it can be labeled and prepared for subsequent studies.
Singota’s R&D Department has been developing a novel microparticle formulation for a large protein molecule. Based on this experience, we can begin to share some of the lessons learned from characterizing the end product.
In the early stages of development, the initial challenge is developing a formulation that consistently delivers a molecule/API that behaves as it’s intended. In our case, we needed a solution containing microparticles in a liquid, which then yielded an elegant powder cake when lyophilized. Microscopic visual inspection was sufficient to tell us when we had successfully encapsulated our molecule. Although in other projects, we would anticipate client specifications to include other, more rigorous analytical tests (e.g., particle size analysis using a differential light scattering system or high-performance liquid chromatography (HPLC) to confirm the integrity and/or quantity of the molecule after encapsulation).
Once you have a molecule/API that consistently passes these initial inspections, the next task is determining whether the product is stable and will have the release profile required at the site of administration.
Stability
A stable product is one that will maintain its useful characteristics from the moment it is placed in storage to the moment it is administered as a drug. For most clients, stability testing involves placing the product in a chamber with controlled temperature and humidity (e.g., refrigerated at 5°C or room temperature, 25°C/60% relative humidity) and then testing samples at specified time intervals.
Various tests involve visual inspection for attributes like color change or time necessary to reconstitute a powder with a liquid like saline. More informative tests measure things like water content (because excess water can denature proteins) and the integrity of the API, using HPLC, capillary electrophoresis (CE), or Fourier transformed infrared spectroscopy (FTIR) to ensure the protein retains its three-dimensional conformation.
The client may require stability testing at regular intervals until the product shows signs of degradation, to gauge its shelf life, or for a specified period (e.g., 2 – 5 years) to ensure that the product will remain stable for its anticipated useful life.
Stability is measured either qualitatively or quantitatively. An example of a qualitative assay would be size exclusion chromatography (SEC), separating compounds in a solution by their size. With our protein, we had a reference standard for the microencapsulated protein and emulsified protein – you expect a characteristic protein spike at a specific retention time; however, each formulation will have its own qualitative assay. A qualitative assay is desirable for protein drugs because a protein’s efficacy may depend on its three-dimensional configuration. To be stable, it must retain its original “folding.”
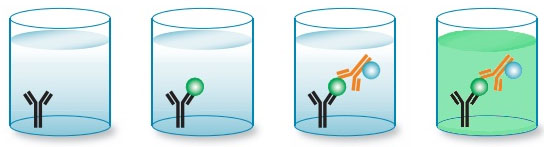
SOURCE: BIO-RAD ANTIBODIES
If you can retain protein with its tertiary structure intact, then you can focus on how much protein is in the solution. Singota’s principal quantitative assay is Enzyme Linked Immunosorbent Assay (ELISA). In ELISA, a sample is placed onto a polystyrene plate seeded with an antibody specific for the protein or API. The antibody holds the API in place and then a second antibody specific for the API is added – the second antibody is tagged with an enzyme. The enzyme catalyzes the reaction with a substrate to produce a green dye. The plate is washed to purge any unbound antibody and then a colorimetric assay (essentially a measure of “how green” the plate appears) provides a measure of how much API is sandwiched between the two antibodies.
There are multiple variations on the ELISA technique and the process needs to be optimized in-house for any given API. Because emulsified systems become complex quickly, a CDMO needs to be able to demonstrate competence in a range of quantitative assays to be effective when working with this type of formulation. It may be unclear which assay to choose, and optimizing each will take significant time. Expect delays and long turnarounds when planning and executing the quantification step of a development microparticle formulation.
Release Profile
The reason for microencapsulating in the first place is to achieve controlled release of the API over time. Specific choices are made during formulation to optimize the pattern of release for a specific site in the body. The drug must endure conditions like temperature and pH along its journey, and it must break apart to release the API when it reaches the target destination.
We test for the desired release profile in vitro, again using ELISA, at defined intervals. We first test immediately upon reconstitution of the drug from its stored form — at that point, we hope and expect to see little or no release of the protein. Over time, we expect to see the release profile increase exponentially (in our experiments, it takes multiple weeks) until it reaches a predictable plateau. After it levels off, the quantity of detectable protein will generally decline due to degradation. Each formulation process will involve experimentation with different methods for reconstitution, including different solvents and different conditions (e.g., pH). Choosing the right reconstitution media takes time and is critical, as it has a direct impact on the percentage of API that is released for assay testing.
Closing Thoughts
Because microparticle suspensions are atypical and diverse, clients with an early-stage microparticle formulation should expect the receiving CDMO to harbor a wealth of analytical equipment. Ideal testing methods and specifications will need to be established early, but chemical interferences and assay complications may quickly lead to a change in plans, especially for quantification. The working relationship between client and CDMO needs to remain flexible to quickly and effectively adjust for these difficulties. In an ideal world, the process would be collaborative and include experimentation and mutual curiosity. In addition, if the project involves a product that is close to preclinical or clinical trial, processes will likely run more smoothly, but all testing would need to be done under GMP conditions, fully validated.
In short, microparticle formulations present multifaceted issues at each step of formulation and characterization. Their many components complicate their creation, storage, and analysis. Any organization looking to undertake a microparticle project, regardless of its stage in the pipeline, should ensure that all affiliated laboratories have adequate equipment and expertise.
Formulation and analytical testing, specifically with microparticles, are both components of Singota’s drug development service capability. For more details on the services, visit our laboratory pages:
