SUS Interchangeability Assessment And Qualification Best Practices
By BioPhorum

Single-use systems (SUS) provide flexibility and speed for delivering medicines to patients. However, supply chain issues have presented significant challenges for biopharmaceutical manufacturers and suppliers throughout the value chain since the coronavirus pandemic.
The lack of clear guidance and standardized criteria for evaluating single-use components (SUCs) has created challenges in responding effectively to supply chain constraints that can disrupt production continuity for suppliers and end users. It also can lead to unexpected deviations when using interchangeable SUCs, introducing performance issues, compatibility problems, and even safety concerns that impede supply continuity. Implementing suitable guidance in this space will help to alleviate some of these issues.
An innovative approach is required to enable end users to remain resilient and sustain the manufacture and supply of medicinal products. Establishing a robust SUS interchangeability program can be a pivotal feature of a supply chain resiliency initiative.
This article provides an industry-aligned framework for suppliers and end users on conducting an equivalency assessment to determine the interchangeability of SUCs based on intended use.
Scope
Our focus is on the use of SUS in drug substance manufacturing. Out of scope are critical SUCs, such as primary packaging, final drug substance/product storage bio-containers, or transport bags. However, the outlined approach may be applied to SUS used in drug product manufacturing, as deemed appropriate by the end user.
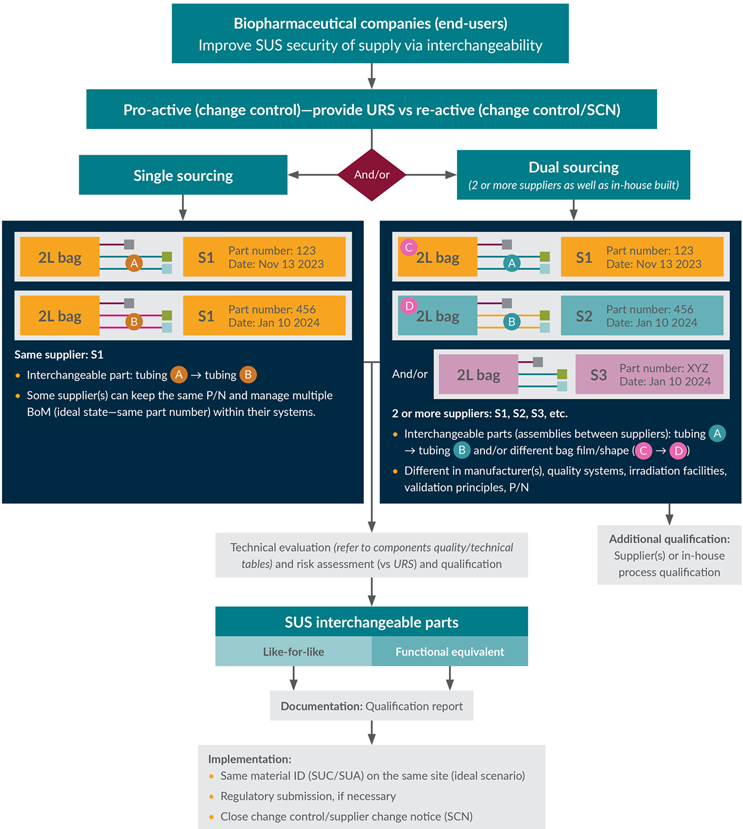
Qualifying an item to be interchangeable with an incumbent item is a prerequisite for dual sourcing. The process below for demonstrating interchangeability focuses on the technical details of comparability and their impact on the product/process. There are scientific and compliance-driven requirements to consider when evaluating two different SUCs or a change to an SUC. As illustrated in Figure 1, the dual-sourcing process is used to achieve SUS interchangeability via a procurement route and is predicated on the result of that material change evaluation. When the dual-sourcing option is taken, other business activities may be required beyond those shown in Figure 1. These may include supplier audits, business/legal agreements, creation of a material identifier, and procurement system setup.
Figure 1: High-level interchangeability business flow chart.
Key: BoM — bill of material, P/N — part number, SCN — supply (or supplier) change notification, SUA — single-use assembly, URS — user requirement specification.
Click on image to enlarge.
Interchangeable Evaluation Methodology
To develop an evaluation framework for interchangeability, careful consideration was given to account for types of SUC change and the variety of assessment inputs/outputs.
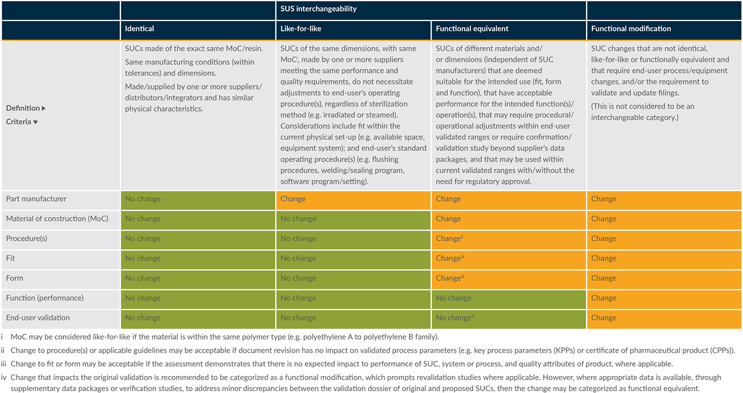
Changes are categorized into four groups (see Table 1 for definitions):
- Identical
- Like-for-like
- Functional equivalent
- Functional modification (not interchangeable)
The four categories represent increasing levels of complexity in the qualification of a candidate for an interchangeable SUC. This structure enhances the agility of the change management process and simplifies the evaluation framework. Strictly speaking, interchangeable SUCs fall into the two middle categories: like-for-like and functional equivalent. To better describe these two categories and facilitate the overall change management process, we have bracketed them with two additional categories: “identical” parts are just that. They need little to no qualification and their assessment should be swift. An example is altering the format of a certificate of analysis. At the opposite end of the spectrum are parts that require “functional modification.” These will require a close to full requalification involving multiple functional groups. This goes beyond the notion of being able to interchange SUCs.
Table 1: Definition and criteria for categories of interchangeability. Click on image to enlarge.
Methodology
The evaluation process focuses primarily on determining the overall impact on fit, form, and function, as well as on validation. It is used to determine whether further supplementary data or assembly qualification is required to deem the candidate SUC (and therefore the candidate assembly or system) acceptable as interchangeable. Other factors are also considered, such as changes to part manufacturer or supplier, which may impact supplier-related qualification activities, and revisions to standard operating procedures/standard work instructions, which could have an impact on validated key process parameters or critical process parameters.
The change categorization framework provided in Table 1 facilitates the evaluation process. The interchangeability methodology and principles outlined are recommended as they cover a comprehensive evaluation approach, including assessing the types of change that may or may not be considered interchangeable. End users are responsible for implementing an evaluation framework that is aligned with their organization’s quality systems.
The terms and criteria in Table 1 are commonly used in industry as part of an assessment to support supplier change notifications. They have been incorporated into this article to support the categorization of applicable changes and are referenced for potential changes to material of construction (MoC), part manufacturer, procedures, and fit, form, and function.
As per Table 1, if the proposed change, assessed against the criteria mentioned above, is found to be identical, then no further technical justification is required. However, where there are differences (e.g., changes in MoC, functional changes), an appropriate technical justification should be provided within the evaluation exercise to demonstrate that there is no impact on the manufacturing process, validation, or compliance dossier. If this cannot be demonstrated, revalidation may be required to qualify the change. Where applicable, the evaluation exercise should focus on criteria such as:
- A change to the MoC may be considered like-for-like when the interchangeable SUC is within the same polymer family (e.g., change from one platinum-cured silicone tubing to another platinum-cured silicone tubing). It assumes similar resins and manufacturing processes, without accounting for differences in additives or catalysts.
- Evaluation of changes to the MoC should determine whether the material is suitable for the process, considering mechanical and physical attributes (e.g., verifying whether the material and its mechanical properties can withstand worst-case process conditions, such as operating pressure and temperature, chemical compatibility, etc.).
- Evaluation of extractables/leachables risk based on change to the MoC: all MoC changes should be risk-assessed in accordance with the end user’s own procedures.
The end user should evaluate common material specifications. Standard attributes applicable to materials typically used in pharmaceutical manufacturing processes have been identified and compiled in Tables 2 and 3 as a guideline to support the assessment.
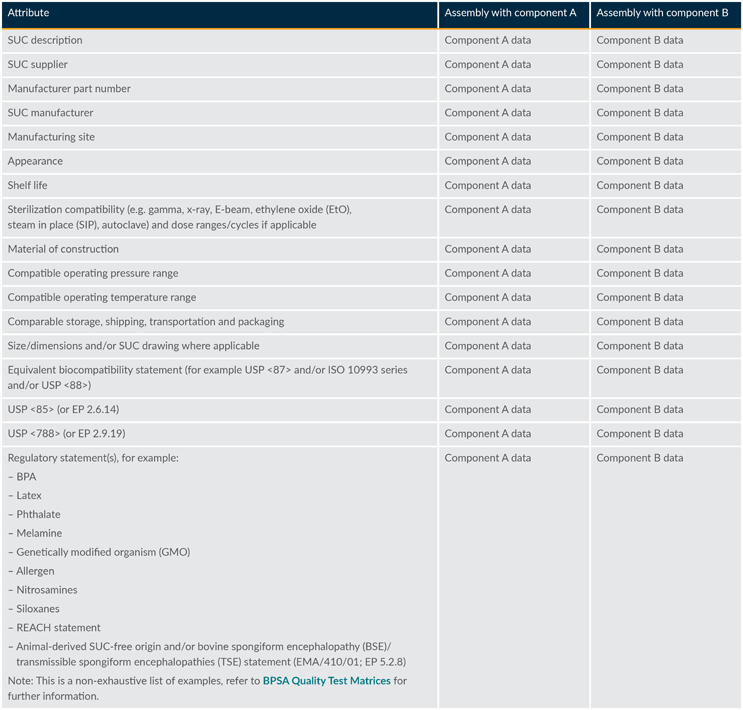
Table 2: Detailed attributes template for fit, form, and function assessment
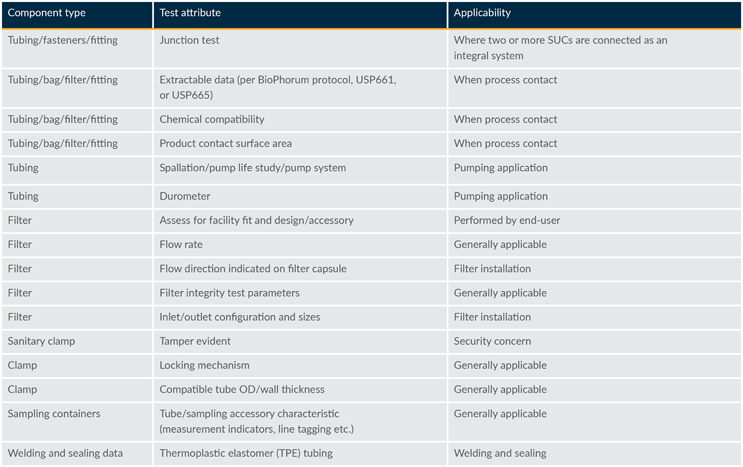
Table 3: Considerations for select materials
The specific attributes used in an assessment will largely be determined by the end user’s requirements specifications. Suppliers are encouraged to use the tables to build robust interchangeability packages for end users to expedite the approval process.
General Considerations
The technical assessment involves a thorough examination of SUC material characteristics and adherence to quality and regulatory standards. A risk-based approach may be taken in accordance with end user procedures. Nonetheless, there are additional factors to consider.
When evaluating prospective interchangeable parts, updates or modifications to supplier systems are necessary to incorporate these changes effectively. The evaluation of supplier system updates focuses on elements such as changes in manufacturer part numbers or stock-keeping unit identifiers.
Suppliers are responsible for providing data packages that meet minimum material evaluation criteria, as outlined in Tables 2 and 3. End users are responsible for assessing the impact on processes and products. If needed, end users should conduct further evaluations and perform any required validation (e.g., filtration or extractables/leachable studies).
Categorizing Impact
On completing the technical evaluation and categorizing the type of change, the category with the highest inherent risk governs the overall change management process. For example, if a change conforms to most criteria within the like-for-like category but requires a verification study to support implementation, the change would then be categorized as a functional equivalent change.
Supply Chain Aspects Of Interchangeability
SUC interchangeability serves as a strategy to protect the supply chain from potential disruptions. Interchangeability efforts may be approached proactively (to protect certain higher-risk items from disruption or to build resiliency into new parts) or reactively (to remediate a known stock-out risk) (see Figure 2).
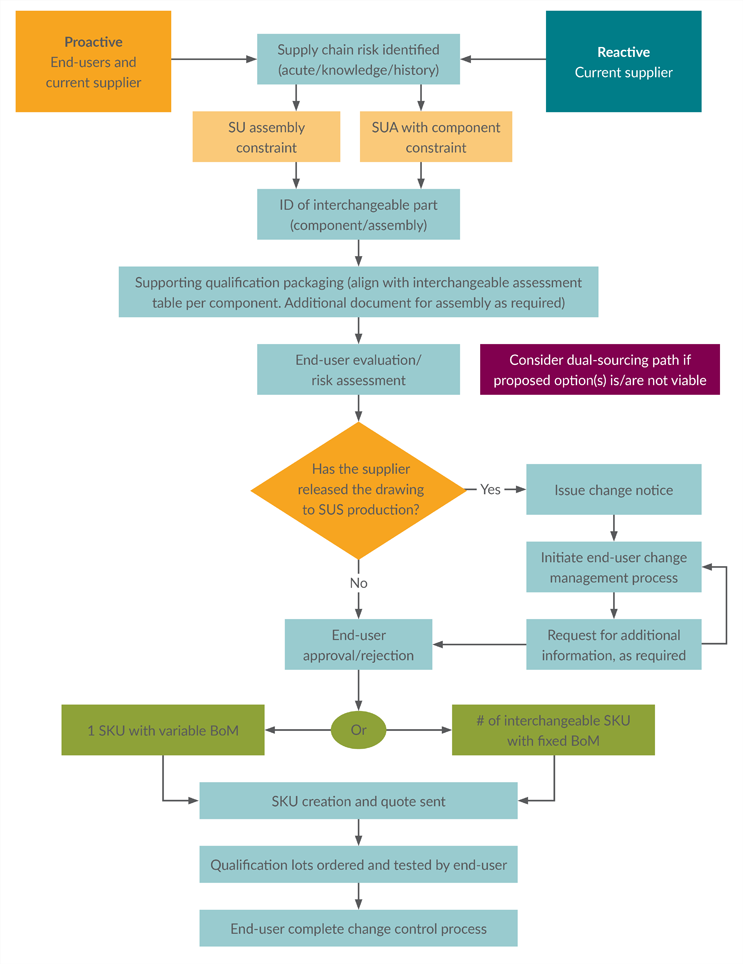
Figure 2: Proactive and reactive interchangeability process flow.
Key: BoM — bill of material, SKU — stock-keeping unit
Interchangeability can be achieved in a single-sourcing solution and/or a dual/multi-sourcing scenario, depending on:
- complexities of the SUS
- options available from the supplier
- risk tolerance of the end user’s process.
Both routes will achieve the goal of interchangeability; the only difference will be in the supply chain route. The evaluation process described above is equally applicable to proactive or reactive approaches.
Reactive And Proactive Interchangeability: Detailed Process Steps
End users and suppliers may choose to employ both proactive and reactive approaches to interchangeability. Although proactive interchangeability requires additional initial up-front investment, it offers long-term benefits by minimizing the need for reactive changes during crises, where shortened timelines often increase the risk of errors.
Both proactive and reactive approaches to interchangeability necessitate qualifying one or more interchangeable SUCs in each assembly or qualifying a second assembly for a given process application.
Understanding Suppliers’ Perspectives
The perspectives shared here highlight the commercial and operational factors to consider to ensure interchangeable SUCs for security of supply. Traceability is reflected in the interchangeable approaches.
This is not an exhaustive list of all approaches, considerations, or impacts that a supplier may encounter when implementing options for interchangeable approaches. There are several approaches that single-use suppliers could use to apply the concept of SUC interchangeability, and various commercial, technical, and operational considerations need to be evaluated.
With catalog SUCs, the supplier has responsibility for providing information to support interchangeability and for ensuring that any substituted SUCs maintain compatibility and mechanical/sanitary properties. Since such changes are often not visible to the end user, the supplier is also responsible for notifying the end user that a change is being made.
For custom SUCs, the supplier and end user should collaborate to define acceptable levels of interchangeability. For example, a supplier may recommend an interchangeable hygienic clamp SUC option using a memo or change notice. This option, once agreed between supplier and end user, could allow the supplier to update the bill of materials for an assembly to use the interchangeable hygienic clamp without updating the assembly technical drawing.
Conclusion
Pandemic-related supply chain constraints forced end users and suppliers to accept and use interchangeable parts under considerable duress, using a myriad of processes and philosophies. From this collective experience, it has become clear that a common risk-based approach to qualifying interchangeable SUCs, rooted in sound science and process understanding, is a prerequisite for maintaining supply chain resilience.
Building on the aggregate experience of end users, integrators, and SUC suppliers, the cornerstone of this best practice is a structured means of categorizing and assessing interchangeability. This is supplemented by discussions on supply chain dynamics, proactive and reactive ways to approach interchangeability, and a supplier perspective on the topic. We recommend that end users take advantage of this information to form the technical basis of their approach to qualifying interchangeable SUCs for single-use bioprocessing assemblies and systems, thereby increasing confidence among internal stakeholders and regulatory agencies.
This article summarizes the main points from a recent BioPhorum publication on this topic. To learn more, check out the full paper, BioPhorum interchangeability assessment and qualification best practice guide and watch the webinar recording on Best practice on the interchangeability of single-use components.