Sterile Formulation And Fill/Finish For Early-Phase Clinical Trials Of Gene Therapy And Biological Products
Source: Ascendia Pharma
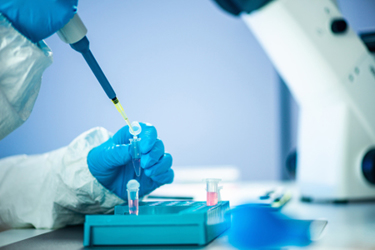
As new gene therapy product development becomes patient targeted and complex, more products are being delivered by injection than orally. Development of parenteral dosage forms is more difficult than oral formulations. Chemical and physical stability are key considerations, viscosity is critical, and sterility is essential.
Ascendia’s team of scientists have decades of combined experience in sterile supplies for clinical trials, making them expert at solving the most difficult formulation challenges. In short, we consistently make the challenge task in biological delivery possible.
access the Brochure!
Log In
Get unlimited access to:
Trend and Thought Leadership Articles

Case Studies & White Papers

Extensive Product Database

Members-Only Premium Content

Welcome Back! Please Log In to Continue.
X
Enter your credentials below to log in. Not yet a member of Outsourced Pharma? Subscribe today.
Subscribe to Outsourced Pharma
X
Subscribe to Outsourced Pharma
