Quantifying Single-Use Waste Produced During mAb Manufacture
By BioPhorum

Considering the healthcare sector’s climate footprint, which accounts for 4.4% of global net emissions, the rising demand for biopharmaceuticals places significant pressure on providers to minimize the environmental impact of these therapies. In this context, the use of plastic is one of several factors to consider when evaluating the environmental impact of drug production (alongside carbon emissions and water usage).
Use of plastic in drug manufacturing has been increasing due to its ability to expedite market readiness. Over the past 20 years, there has been a shift from producing biopharmaceuticals in traditional stainless-steel facilities to employing single-use technologies (SUTs), which entail high-quality plastic systems with numerous complex interconnected components. A primary goal of this transition has been to lower capital costs and accelerate time-to-market.
The rise in chronic diseases across populations, coupled with the resultant forecast growth of the biopharmaceutical sector, suggests an increasing amount of associated SUT plastic waste.
Although the scope of the products included in that estimation is not outlined in the article, it provided a perspective on the possible scale of the issues to address and the need for a better understanding of the nature of waste material to explore improved waste management streams.
When we considered the scope of single-use plastics used in biopharmaceutical production, we identified four distinct domains (see Figure 1).

Figure 1: Four domains of single-use plastics in biopharmaceutical production
This article shares data and insights on the current scale of plastic SUT waste and its projected growth, specifically for the manufacturing domain. It encourages industry leaders across the biomanufacturing value chain and the recycling community to explore innovative methods to reduce plastic waste volumes and retain some of this material’s value through recycling.
Quantification Of SUT Plastics Waste
The data presents a comprehensive and transparent quantification of SUT waste generated during the manufacture of therapeutic proteins, with a specific focus on commercial-scale monoclonal antibodies (mAbs) in the manufacturing domain only.
It is crucial to recognize that, as only one in every 10 therapies advances from clinical development to commercial production, the clinical development phase of production may involve substantial material consumption. While other modalities (e.g., vaccines) and the SUT plastics used at other stages of production (e.g., the clinical phases of mAb development) are not directly included in this study, the findings remain applicable and informative. By quantifying the volumes of SUT plastic waste associated with commercial mAb production, we aim to establish a data baseline that the industry can use to guide future sustainability efforts and improvements.
To quantify plastic usage associated with a typical commercial-scale mAb manufacturing process, a detailed process model has been developed. See Figure 2 for an outline of the process flow.
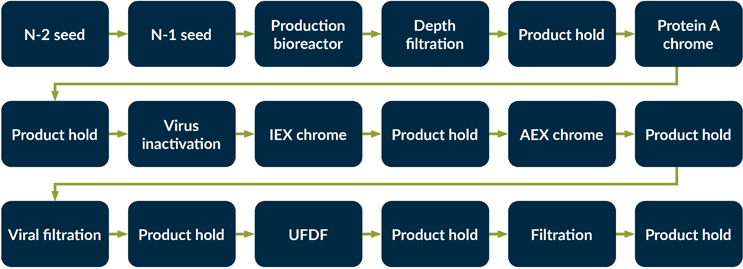
Figure 2: Process flow for the mAb manufacturing process.
Key: AEX — Anion exchange chromatography, IEX — Ion exchange chromatography,
UFDF — Ultrafiltration and diafiltration
The plastics information incorporated into the process model is based on substantial quantified SUT weight data from suppliers and end users, which has been aggregated and blinded. Waste material from these SUT products is generated by license holders and CDMOs following production of the therapeutic product. Although this waste constitutes a relatively small proportion of the overall plastic waste along the value chain, the materials are valuable, high-specification polymers that, due to the complex nature of the multi-polymer films and the hazardous classification of the waste, are currently lost to landfill or incineration. The scope of the products quantified in this study is highlighted in Figure 3.

Figure 3: Manufacturing plastics — SUT products quantified in this study
Two manufacturing scales have been considered: large-scale stainless steel (12,000 L) and intermediate-scale single-use (2,000 L). The impact of varying product titer (2 g/L, 5 g/L, and 10 g/L) and buffer preparation philosophy has been considered to support an evaluation of the sensitivity of the results.
The results provide a breakdown of plastic waste generated on a per-batch basis. The per-batch results have been used to estimate the annual SUT plastic waste quantities associated with mAb manufacturing based on published market trend data for the industry.
Key model assumptions include:
- Results are presented on a per-batch basis with a single bioreactor and downstream train considered for each batch. A 70% product yield is assumed in all cases.
- Single-use assemblies associated with single-use bioreactors, single-use product mixers, and single-use buffer/media preparation and hold systems (which represent a high proportion of regular plastic waste) have been included within the scope of this study. The model does not consider ancillary plastic-containing items, such as single-use filters, single-use tubing, chromatography resins, etc. Therefore, the output is not a total quantification of all solid waste generated by the manufacturing process.
- Assembly weights based on single-use product data from suppliers and end users have been considered (see Figure 4), provided by single-use component suppliers and manufacturing companies. Assembly weights include packaging, which accounts for 30%-40% of the stated weight.
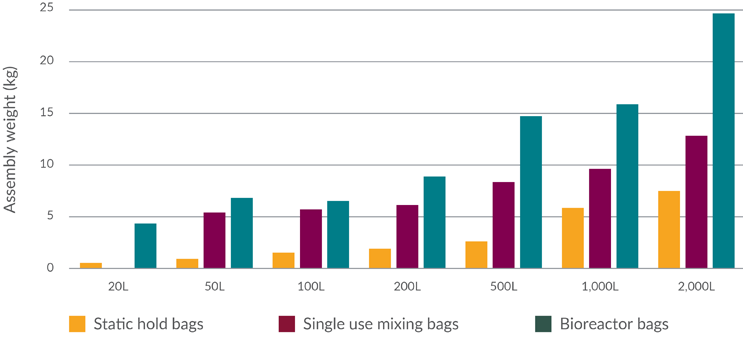
Figure 4: Single-use assembly weights (including packaging) — median results
Results: Single-Use Plastic Waste — Per Batch
The quantity of single-use plastic waste generated per batch for intermediate and large-scale manufacturing is outlined in Figure 5.
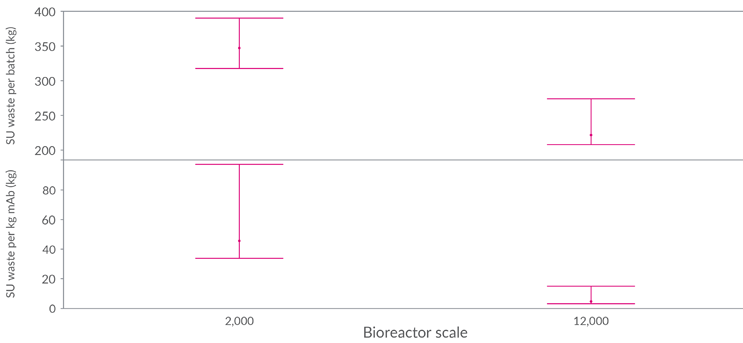
Figure 5: Single-use plastic waste on a per batch (top)/kilogram mAb basis (bottom) for 2,000 L and 12,000 L manufacturing scale (weighted mean considering all variables with 95% confidence interval)
As expected, the quantity of single-use plastic waste for a 2,000 L process is much higher than for a 12,000 L stainless steel process. Taking a 2,000 L process with a 5 g/L product titer (using traditional buffer preparation), 369 kg of plastic waste is generated per batch. In the same scenario, at 12,000 L scale, the plastic waste per batch is significantly lower at 186 kg, because a substantial proportion of the process needs exceeds the considered single-use threshold.
While absolute quantities of plastic waste in large-scale manufacturing are lower, they remain substantial with SUT being used for seed bioreactors, smaller-scale media, and buffer preparation and hold.
Results: Breakdown Of Single-Use Plastic Waste — By Facility Area
A breakdown of single-use plastic waste by area is provided in Figure 6.
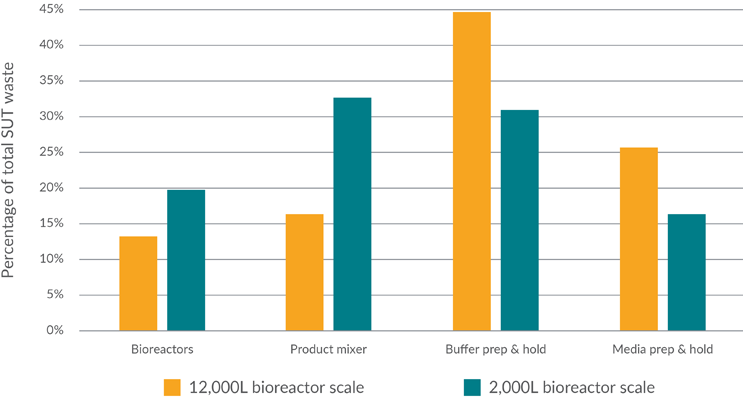
Figure 6: Breakdown of single-use plastic waste by area of biopharmaceutical manufacturing process
Considering all variables (bioreactor scale, product titer, and buffer preparation philosophy), the core process (bioreactors and product hold) accounts for 52% and 30% of the total waste at the intermediate and large scales, respectively, with media and buffer providing the remaining 48% and 70%.
The breakdown of waste is heavily influenced by buffer preparation philosophy. Taking a 2,000 L-scale process with a 5 g/L product titer (using traditional buffer preparation), the proportion of waste generated by media and buffer increases to 53% when compared with the overall average of 48%. An equivalent scenario at the 12,000 L bioreactor scale has media and buffer accounting for 72% of overall waste. Given the continued prevalence of traditional buffer preparation in the industry, these figures are likely closer to the current industry state.
As buffer is the largest constituent by volume in manufacturing, it understandably represents a large proportion of plastic waste, representing 31% of SUT waste at an intermediate scale and 45% at a large scale. This is particularly relevant in the context of recycling, as buffer preparation and hold systems offer significant potential, given the lack of biological contamination.
Sensitivities Of Plastic Waste Volumes To Process Variables
The absolute quantity of plastic waste generated by a manufacturing process is highly variable and dependent on product parameters, manufacturing technology, and operational strategy. The significant influence of buffer management philosophy is outlined in Figure 7.
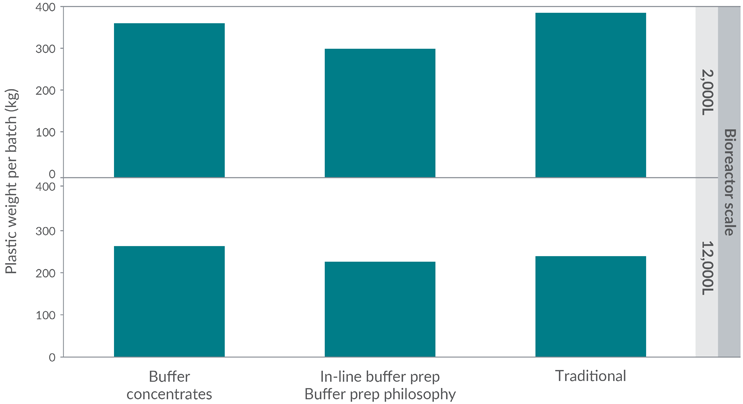
Figure 7: Influence of buffer management philosophy on waste quantity at 2,000 L (top) and 12,000 L (bottom) production scale
The use of advanced buffer management strategies (such as buffer concentrates and in-line buffer preparation) may increase plastic waste associated with buffer at a large scale and decrease it at an intermediate scale. However, in all scenarios, the quantity of waste associated with buffer remains substantial.
At a large stainless-steel scale (12,000 L), the use of advanced buffer manufacturing strategies increases the potential for using SUTs as both buffer concentrates and in-line buffer preparation reduce the volumes of liquids to be handled, such that a greater proportion fits within the volumes suitable for single-use technology.
The use of buffer concentrates in particular increases demand for single-use items. However, buffer stock blending (or equivalent) reduces the absolute number of preparations (and storage systems) needed as well as volumes, such that the quantity of plastic waste generated is lower (compared to buffer concentrates), even though more stainless-steel systems can be replaced with single-use technology.
Manufacturing SUT Waste At A Global Level
The data in this article can be used to provide an order of magnitude of the quantity of waste produced by industry annually (for the components included in the scope of this study). For example, , an estimated 25,000 kg of mAbs were produced in 2019. Based on this figure, the associated SUT plastic waste in the scope of this study would have been 352,000 kg (based on median plastic weights). When other modalities are considered, as well as noncommercial manufacturing (e.g., clinical scale), the actual quantity of SUT plastic waste from the production of biologics for 2019 would likely have been considerably higher. Some estimates projected that total mAb production would have risen to ≈56,000 kg by 2024, which would represent a doubling of single-use plastic use since 2019. The correlation between potential increased industry output and single-use plastic waste is outlined in Figure 8.
While numbers for industry output are difficult to estimate, the general trend toward growth is clear and this will result in a greater demand for SUT, a correspondingly greater amount of waste, and an increasing need for sustainable solutions.
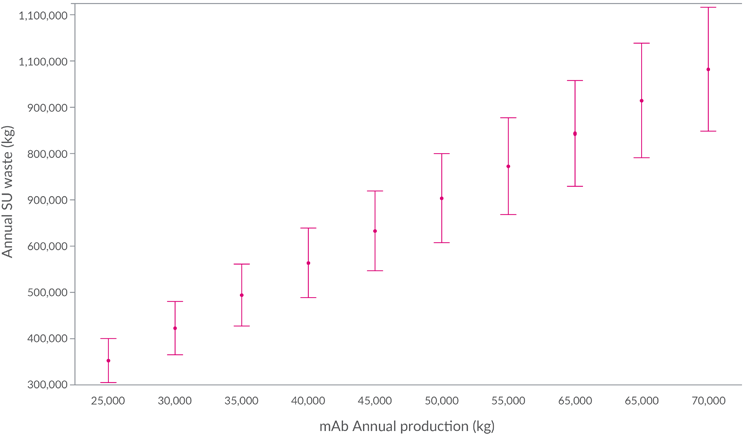
Figure 8: Estimated annual single-use plastic waste based on the corresponding annual mAb demand
Conclusion
The detailed quantification data of plastic waste generated in the manufacture of mAbs on a batch basis provides a comprehensive evaluation of the breakdown of SUT manufacturing waste within a facility, which can be used to support future recycling initiatives.
A greater understanding of the waste source within a facility will be pivotal for future sustainability improvements and will demonstrate the importance of considering process support areas, such as media and buffer, in addition to the core process.
License holders can use the model to quantify their waste volumes at each site, which will help build a regional picture and enable solutions to be developed with waste management solutions providers for specific geographies (we note that solutions are already available in specific regions).
The impact of process developments (e.g., increasing product titer) and manufacturing strategies (e.g., buffer preparation philosophy) on overall plastic waste quantities has been demonstrated. As the industry becomes more efficient with higher-titer processes, plastic usage will increase accordingly, particularly at the intermediate scale. While SUT has been demonstrated to offer environmental advantages over traditional stainless steel, there remains a responsibility to minimize unnecessary waste. The complex nature of this waste poses challenges for recycling. However, the high-value plastics used in SUTs and the resulting quality of the waste material offer opportunities for innovative solutions that may embed circularity in the design of products, enhance recycling capabilities, and reduce reliance on virgin material extraction.
By collaborating and providing better data access, biomanufacturers, their suppliers, and waste management solution providers can develop more sustainable methods for managing valuable plastics, enhancing circularity, and preserving material integrity so that global communities continue to reap their benefits.
This article summarizes the main points from a recent BioPhorum publication on this topic. To learn more, check out the full paper, Estimating the volume of single-use waste produced during drug substance manufacturing of monoclonal antibodies. It includes detailed appendices on single-use assembly weights, the quantity of single-use assemblies required, and the quantity of plastic waste generated for each considered scenario on a per-batch basis.
