Nucleoside Triphosphates (NTPs) Emprove® Expert
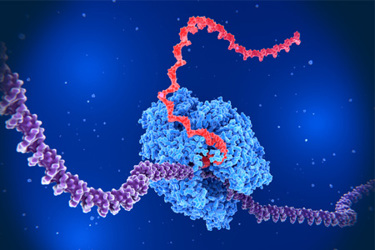
High‑purity nucleoside triphosphates are essential for reliable mRNA production. Examine a portfolio designed to meet the stringent expectations of modern therapeutic and vaccine manufacturing. Learn more about the role of both natural and modified NTPs in IVT processes, along with key physicochemical specifications such as ≥99% purity, sodium‑salt formulation, and consistent 100 mM concentration, as well as get a clear view of the quality controls behind these materials—including nuclease‑free processing, low endotoxin and microbial limits, and expanded impurity testing—to support high‑risk biopharma applications.
For teams focused on mRNA workflows, see how the Emprove® regulatory support framework streamlines compliance to help ensure process consistency and regulatory readiness.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Outsourced Pharma? Subscribe today.
