Integrated Single-Site CDMO Model
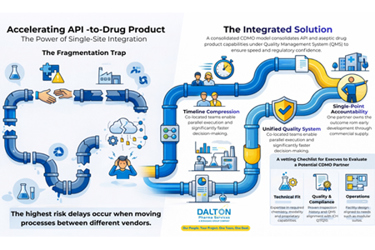
Fragmented outsourcing often slows drug development, creating delays at vendor handoffs, increasing documentation gaps, and raising contamination and compliance risks. A single‑site approach consolidates development, manufacturing, and aseptic fill‑finish operations under one coordinated quality framework, reducing these friction points and improving predictability. By eliminating repeated tech transfers, organizations preserve process knowledge, streamline qualification activities, and accelerate issue resolution through close collaboration among co‑located teams. This structure aligns tightly with modern regulatory expectations, particularly those emphasizing contamination control, data integrity, and unified oversight.
Integrated operations also shorten the path from API development to clinical supply by enabling parallel workstreams and reducing the need for rework later in the program. A single quality management system supports cohesive change control, trending, and governance, while modular facility design helps maintain flexibility and capacity. For early‑stage and high‑risk programs, this model offers a lower‑risk, inspection‑resilient path that balances speed, consistency, and technical rigor across the entire development lifecycle.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Outsourced Pharma? Subscribe today.
