India To Enhance Oversight Of Medical Devices
By Gunjan Bagla, Amritt, Inc.
 India's Central Drugs Standard Control Organization (CDSCO), the country’s national regulatory authority for both drugs and medical devices (i.e., India's equivalent of the United States’ FDA) is acting on a report submitted by its internal Drugs Technical Advisory Board (DTAB) to form a new division under the CDSCO.
India's Central Drugs Standard Control Organization (CDSCO), the country’s national regulatory authority for both drugs and medical devices (i.e., India's equivalent of the United States’ FDA) is acting on a report submitted by its internal Drugs Technical Advisory Board (DTAB) to form a new division under the CDSCO.
According to the proposal by DTAB — the country’s highest statutory decision-making body on technical matters related to medicines and medical devices — the new division and its officers will work exclusively with medical devices. Critical care devices, such as coronary stents, pacemakers, orthopedic knee and hip implants, structural heart devices, peripheral devices, endoscopes, and in vitro diagnostic devices, are expected to be prioritized.
Fig. 1 — The new division’s structure, as proposed by DTAB
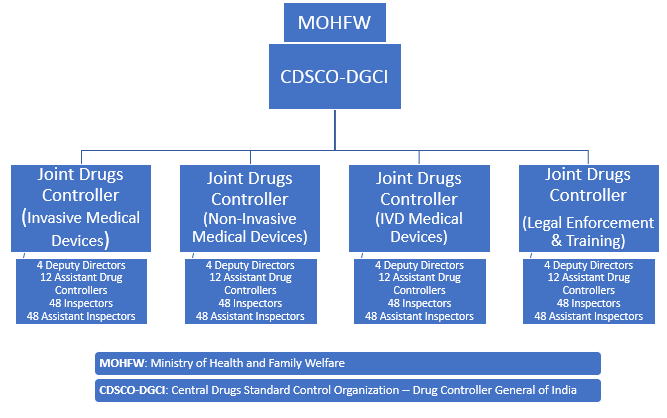
India hopes to hire physicians and surgeons across therapeutic segments covering orthopedics, dermatology, surgery, biomedical engineering, medical imaging, cardiology, and biocompatibility, to name a few. Additionally, five laboratories will be established to test devices and in-vitro diagnostics. Their locations have not yet been disclosed.
Over 700 personnel, including administrators and subject matter experts, will be hired for the new division over time. DTAB determined this number in light of the workload that regulators will need to undertake to streamline regulations, monitor the efficacy and quality of the medical devices in India, and strengthen this vertical.
More than 400 of the 700 proposed staff are expected to oversee regulatory tasks, such as audits at manufacturing sites, import approvals, cancellations, and approval of licenses, among other duties. The remainder of new hires will be deputed at national laboratories to test the safety and efficacy of the medical devices as proposed by the manufacturers during the submissions.
Table 1 — Proposed Vertical for Medical Devices in CDSCO under DCGI

Total number of personnel = 754
In addition to the above personnel, DTAB also has proposed to recruit some experts on a contractual basis for the following categories:
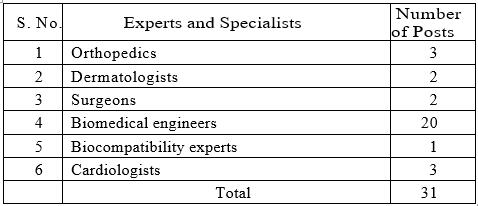
The expectation is that CDSCO’s talent search will primarily focus on domestic candidates. However, due to U.S. visa issues, more Indian expats are returning to India, and some of them may find these opportunities interesting. The government will be attracted to such American-trained candidates.
Industry experts feel that the full infrastructure for this new division might take over five years to create and sustain. The current resource strength at CDSCO is not even half that number.
Foreign companies selling into India generally welcome the sharpened focus on medical devices as distinct from drugs, as pre-existing “catch-all” regulations in India have failed to properly regulate many device classes. Indian and some Chinese companies have jumped into this void with products that may or may not be safe and effective, but most Western makers prefer to have a proper approval from the government before marketing any product.
This has caused some Western companies to refrain some entering certain categories. Their hope is that the increased regulation and inspections will shake down marginal operators who might not always follow international standards such as ISO 13485, Six Sigma quality, and Good Manufacturing Practice.
EDITOR NOTE: Shortly before publication of this article, India announced an unexpected and deep cut in corporate tax rates -- amounting to roughly $20 billion in reductions at the current size of the economy. Combined with the described regulatory changes, this move has the potential to heavily impact both current and future medtech operators in the country.
The government slashed the corporate tax rate for Indian companies to 22 percent from 30 percent with effective tax rate lowered to 25.2 percent from 34.9 percent earlier (including surcharge and cess) and cut the minimum alternate tax rate to 15 percent from 18.5 percent. The government also announced a special 17 percent rate for new companies incorporated on or after Oct. 1, 2019, and starting new manufacturing facilities before March 2023.
By Monday (9/23/2019) evening, the “Sensex” (similar to the Dow Jones average) had risen over 3,000 points, and overall stock values were up about 8 percent, marking the best two-day performance the Indian stock market has ever seen.
Finance Minister Nirmala Sitharaman said on Sunday (9/22/2019) “The top consideration on which India was rejected as an investment destination is now better than everybody else… For someone who is coming up with new investment, no country is offering 15 per cent (base tax rate, effective 17 per cent). We are giving 15 per cent with no MAT (minimum alternate tax) and simpler taxation structure.”
You can find the author's original article on the tax change here.
About The Author
Gunjan Bagla is managing director of Amritt Inc., a California-based consulting firm focused on helping American companies to succeed in India. His clients include Covidien (now Medtronic), Roche Diagnostics, BD, Lifenet Health, Johnson & Johnson, Gojo, and more. He has appeared in The New York Times, the Los Angeles Times, and the Washington Post, as well as on Bloomberg TV, BBC Television, and Fox Business News. He also writes about India for the Harvard Business Review. Gunjan has an MBA from Southern Illinois University and a mechanical engineering degree from the Indian Institute of Technology (IIT) Kanpur in India.
Resources
http://www.theindiaexpert.com/india-to-streamline-regulatory-oversight-of-medical-device
